Short Case Summary
74 years old smoker, male patient presented with significant weight loss, productive cough and abdominal discomfort of short duration. Patient was icteric with stable vitals. On examination of chest, there was decreased air entry over right lung field. On examination of abdomen, liver was palpable with hard, nodular surface.
On investigations:
CBC: Anemia (9 gm/dl Hb), otherwise normal
LFT:
- Total bilirubin: 6.6 mg/dl
- Direct bilirubin: 3 mg/dl
- ALT: 280 U/L
- AST: 460 U/L
- ALP: 1500 U/L
RFT, Urine R/E and HIV/Hepatitis serology: Nothing abnormal
USG abdomen/pelvis revealed:
- Hepatomegaly with heterogenous parenchyma with numerous ill-defined nodules.
- Ascites +
Tumor markers:
- CA 19-9: 385 U/ml
- CEA: 290 ng/ml
- Alpha-fetoprotein: 4.7 ng/ml
FNA of liver: was consistent with metastatic small cell carcinoma
CECT chest showed:
- Irregular heterogenously enhancing central right lung mass (55 mm in largest diameter) with collapse-consolidation of right anterior segment of upper lobe.
- Enlarged right paratracheal, pre-carinal and sub-carinal lymph nodes.
- Small right pleural effusion.
- Numerous poorly enhancing hypodense lesions in liver – suggestive of metastatic nodules.
Clinical Diagnosis: Right sided small cell carcinoma (HPR: small cell carcinoma) with multiple lymph node involvement with right sided pleural effusion with multiple liver metastases T2bN2M1b; Stage IV/Extensive or Metastatic disease.
Case Discussion
Small Cell Lung Cancer (SCLC)
Incidence in Western World: Dropped to 13%
Association: Smoking habits
Origin: Neuroendocrine-cell precursors
Characteristics: Rapid growth and high response to both chemotherapy and radiotherapy
Location: Frequently central
Site of biopsy for pathological diagnosis: A biopsy from a metastatic lesion may be the preferred option if the location of the metastasis is easily and safely accessible to biopsy, as this will also pathologically stage the patient (e.g. liver, skin). Bronchoscopy may also be preferred due to the central location of the tumor in other cases.
Immunohistochemistry to confirm the diagnosis of SCLC (synaptophysin, chromogranin A, CD56, thyroid transcription factor 1 and MIB-1) is not mandatory.
Staging and risk assessment:
- Initial assessment should include smoking history, physical examination, complete blood count, liver enzymes, sodium, potassium, calcium, glucose, lactate dehydrogenase levels and lung (if localised disease) and renal function tests.
- A computed tomography (CT) scan with contrast of the chest and abdomen is recommended.
- In localised disease or if symptoms or clinical findings suggest involvement, additional bone scintigraphy and CT or MRI of the brain are recommended.
- 2-fluor-2-desoxy-D-glucose positron-emission-tomography (FDG-PET CT) scan is optional in localised disease. PET findings, which modify treatment decisions, should be pathologically confirmed.
- A bone marrow aspiration and biopsy should be carried out in the case of abnormal blood counts suggesting involvement, particularly in localised disease.
- Version 7 of the TNM staging system according to the Union for International Cancer Control (UICC) should be used. TNM staging of lung cancer has been simplified already in the previous article.
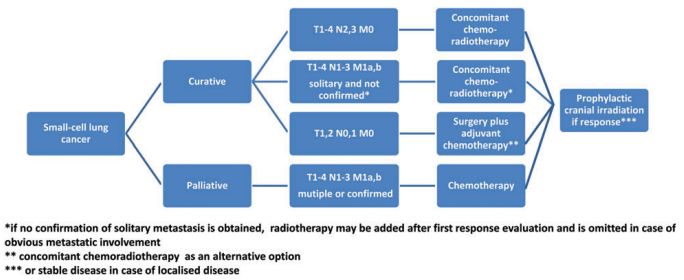
Algorithm for Small Cell Lung Cancer Management:
Localized disease (T1-4, N0-3, M0):
Median survival and 2-year survival rates have been reported to be 15–20 months and 20%–40% respectively. Importantly, the proportion of patients who survive for 5 years has been reported to be 20%–25%.
T1-2 N0-1 M0:
- 5% present at this stage.
- Favourable outcomes with 5-year survival rates in the order of 50%.
- A surgical approach in this group of patients is justified after ruling out mediastinal lymph node involvement (i.e. negative lymph nodes on CT scan, PET-CT scan or EBUS and/or mediastinoscopy if enlarged). Postoperatively, 4 cycles of adjuvant chemotherapy should be administered.
- In the case of unforeseen N2 or N1 or in patients who have not undergone systematic nodal dissection, postoperative radiotherapy should be considered.
- Combined concurrent chemoradiotherapy is an alternative treatment.
Other patients with localized disease:
- Treat with: Concurrent chemotherapy and thoracic radiotherapy.
- Starting chest radiotherapy within 30 days after the beginning of chemotherapy is preferred.
Metastatic disease (M1):
Treatment of Stage IV, metastatic disease is palliative combination chemotherapy. Various combination chemotherapy regimens demonstrate similarly high response rates (RRs) of 60%–70%. Due to frequent rapid relapse and limited activity of second-line treatment, overall survival (OS) remains poor (<10 months).
1st line of treatment:
- 4–6 cycles of etoposide plus cisplatin or carboplatin.
- In young patients and patients with localised disease, etoposide–cisplatin is recommended.
- Irinotecan–cisplatin, gemcitabine–carboplatin (in poor prognostic patients only) and i.v. or oral topotecan–cisplatin are alternative options if etoposide is contraindicated.
2nd line of treatment:
- Oral or i.v. topotecan are recommended for patients having resistant or sensitive relapse with CAV (Cyclophosphamide, Doxorubicin/Amrubicin, Vincristine) being an alternative option.
- Patients with sensitive relapse may derive benefit from reintroduction of the first-line regimen (usually platinum– etoposide).
All Small cell lung cancer patient should be considered for prophylactic cranial irradiation (PCI) if they have responded to initial treatment.
Follow-up and long-term implications:
- Probability of second malignancy in continued smoking warrants smoking cessation counselling.
- 2-3 monthly CT scans in Metastatic disease.
- 6 monthly CT scans for 2 years with lengthening of intervals thereafter are recommended for patients with nonmetastatic disease who have received potentially curative treatment.
Source: ESMO Clinical Practice Guidelines
Etiopathogenesis and Paraneoplastic syndromes in bronchogenic carcinoma has already been discussed earlier.
Chemotherapy Regimens
The following treatment recommendations should be given for a maximum of 4-6 cycles:
- Cisplatin 60-80 mg/m 2 IV on day 1 plus etoposide 80-120 mg/m 2 IV on days 1-3 every 21-28d or
- Carboplatin AUC 5-6 IV on day 1 plus etoposide 80-100 mg/m 2 IV on days 1-3 every 28d or
- Cisplatin 60 mg/m 2 IV on day 1 plus irinotecan 60 mg/m 2 IV on days 1, 8, and 15 every 28d or
- Cisplatin 30 mg/m 2 IV on days 1 and 8 or 80 mg/m 2 IV on day 1 plus irinotecan 65 mg/m 2 IV on days 1 and 8 every 21d or
- Carboplatin AUC 5 IV on day 1 plus irinotecan 50 mg/m 2 IV on days 1, 8, and 15 every 28d or
- Carboplatin AUC 4-5 IV on day 1 plus irinotecan 150-200 mg/m 2 IV on day 1 every 21d or
- Cyclophosphamide 800-1000 mg/m 2 IV on day 1 plus doxorubicin 40-50 mg/m 2 IV on day 1 plus vincristine 1-1.4 mg/m 2 IV on day 1 every 21-28d.
2nd line regimen for metastatic disease:
- Etoposide 50 mg/m 2 PO daily for 3wk every 4wk or
- Topotecan 2.3 mg/m 2 PO on days 1-5 every 21d
Source: Small Cell Lung Cancer Treatment Protocols | Medscape
Calveart formula is used to calculate dose of Carboplatin:
Dose adjustment of Chemotherapeutic agents in liver dysfunction
Chemotherapeutic agents whose dose adjustment is not required in mild-moderate liver dysfunction:
- Platinum agents: Carboplatin, Cisplatin. Oxaliplatin
- Capecitabine, 5-fluorouracil
- Cyclophosphamide
- Mechlorethamine
- Topotecan
Recommended dose adjustments for liver dysfunction:
Source: Chemotherapy dosing in the setting of liver dysfunction | Cancer Network
Liver Enzymes in Hepatic Metastases
Liver Function Test (LFT) may fail to detect significant number of patients with liver metastases. In a study, 20-33% had normal levels of all 3 liver enzymes (ALP, AST, ALT) within 2 weeks of detection of liver metastases by CT scan. 1
Compared to viral hepatitis and drug-induced liver damage, where AST and ALT are raised to the same degree, metastatic lesions of the liver presents with rise in AST more than ALT like in Alcoholic liver disease.2
Jaundice in the face of a high platelet count is suggestive of acute liver disease or metastatic cancer/lymphoma involving liver.
ALP may be raised in both the bone and hepatic metastasis. However, the ALP from bone is heat labile, while the ALP drom liver is heat stable.
Alpha-fetoprotein in Hepatic Metastases
Alpha-fetoprotein is not a marker for liver metastases but the priamry hepatocellular carcinoma (HCC). However, few cases have been reported with rise of AFP in liver metastases but these often resulted from the AFP-secreting tumor outside the liver like germ cell tumors, AFP-secreting gastric or colo-rectal cancer. These entity should be considered in patients with liver lesions and an elevated AFP without risk factors for hepatocellular carcinoma.
It is postulated that maturation arrest of liver-determined tissue stem cells give rise to hepatocellular carcinomas. Alpha-fetoprotein (AFP) is a glycoprotein produced by the fetal liver and yolk sac during pregnancy. AFP is not raised in all cases of HCC and may also be raised in benign liver conditions. In general, consistently elevated serum AFP levels greater than 500 ng/mL are indicative of HCC.3 A high AFP concentration ≥ 400μg/L in HCC patients is associated with greater tumour size, bilobar involvement, portal vein invasion and a lower median survival rate.4 AFP-L3 is produced by malignant liver cells. Levels >1000 ng/ml may indicate Hepatoblastoma.
Elevated CEA in Small Cell Carcinoma of Lung
Carcinoembryogenic antigen (CEA), lactic dehydrogenase (LDH) and ferritin have been studied in patients with SCLC. NSE (Neuron specific enolase) has been widespreadly used in management of patients with SCLC and CEA, ferritin and LDH are valuable, easily and routinely used than many tumor markers.
A study concluded that, there was no relationship of CEA level to the site of metastases in small cell carcinoma of lungs, although levels greater than 100 ng/ml were always associated with liver metastases.5
In a prospective study of 116 patients, CEA was found to be the most frequently detected tumor marker in SCLC, with 32.8% of samples scoring positive, and was also the only marker that correlated with overall survival. The average survival time was 16.78 months for patients with CEA below 5 ng/ml and 11.4 months for patients with CEA above 5 ng/ml.6
Elevated CA19-19 in Small Cell Carcinoma of Lung
Increased serum levels of CA 19.9 in lung tumors, especially in adenocarcinomas, are a known fact, although uncommon. CEA and CA 19-9 have been associated often with Non-small cell lung cancer than Small cell lung cancer. In a letter written by José María Prieto De Paula et.al. to the editor of Arch Bronconeumol, they deduced that CA 19.9 may present higher levels in small-cell lung cancer, although its determination is not considered clinically useful.
However, cholestasis is believed to play an important role in causing raised CA 19-9 level. Hence, caution is needed in interpreting elevated CA 19-9 in patients with jaundice. However, the tumour marker was less than 100 D/ml in cases of hepatitis in a study.7
In cases of colorectal carcinoma, it has been found that CEA is associated with extent of liver metastases and CA 19-9 is associated with the multiplicity of the liver deposits.
Pleural effusion in Small Cell Carcinoma of Lung
In a study, around 20% patients with small cell lung cancer were found to have minimal pleural effusion and 23% were found to have malignant pleural effusion and the presence of effusion was associated with worse survival and should be considered when CT scans are interpreted. The underlying mechanism is most often due to the mediastinal lymphadenopathy.8
Metastatic sites in Small cell carcinoma of Lung
Retrospective study conducted by Kensuke Nakazawa et.al., the incidence of distant metastasis at the time of initial diagnosis of SCLC in this study was 60.6%, and the most common metastatic sites were the liver (20.3%), bone (18.3%), brain (15.5%), lung (10%) and adrenal glands (6%). They also suggested that existing liver, bone and brain metastasis adversely affects the outcome of the disease.9
Study conducted by M.Riihimaki et.al. concluded that the liver (35%) and nervous system (47%) metastases were common in patients with metastases from small cell lung cancer. Younger age at diagnosis was associated with increased metastases, except for metastases to respiratory organs and the liver. Women more frequently had metastases to the nervous system. Survival in metastatic lung cancer was worst in patients with liver or bone metastases, and better in nervous system metastases.10
Primary small cell carcinoma of Liver
The most common site of small cell carcinoma is the lung. It has rarely been found at extrapulmonary sites such as the trachea, larynx, thymus, esophagus, stomach, small intestine, colon, prostate, gallbladder, skin, breast, and uterine cervix. Small cell carcinoma, involving primarily the liver, is extremely rare. Extrapulmonary small cell carcinoma of the liver is histologically indistinguishable from metastatic pulmonary small cell carcinoma to the liver. Therefore, exclusion of pulmonary small cell carcinoma is a prerequisite for the diagnosis of extrapulmonary small cell carcinoma.
In most reported cases, the tumor showed neuroendocrine differentiation, with markers like enolase, synaptophysin and chromogranin. Most cases also reported CD56, thyroid transcription factor-1 (TTF- 1), c-kit, cytokeratin, and CEA. However, it is not necessary to find all markers in every case. Presence of vimentin, desmin, CD56, chromogranin, synaptophysin, and S-100 protein is quite specific. Synaptophysin was positive in 100% of the cases and neuron specific enolase was positive in over 85% of the cases.11