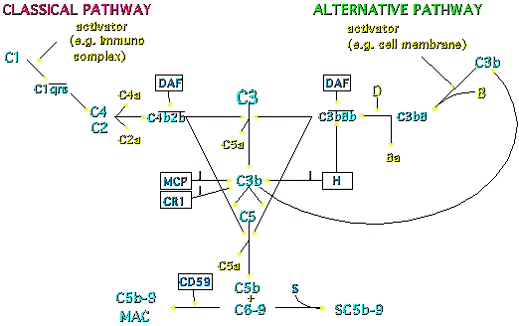
The complement system is composed of about 20 different proteins released into the blood after production in the liver. They interact in coordinated and regulated way to produce biologically active protein products.
ACTIVATION OF COMPLEMENT CASCADE
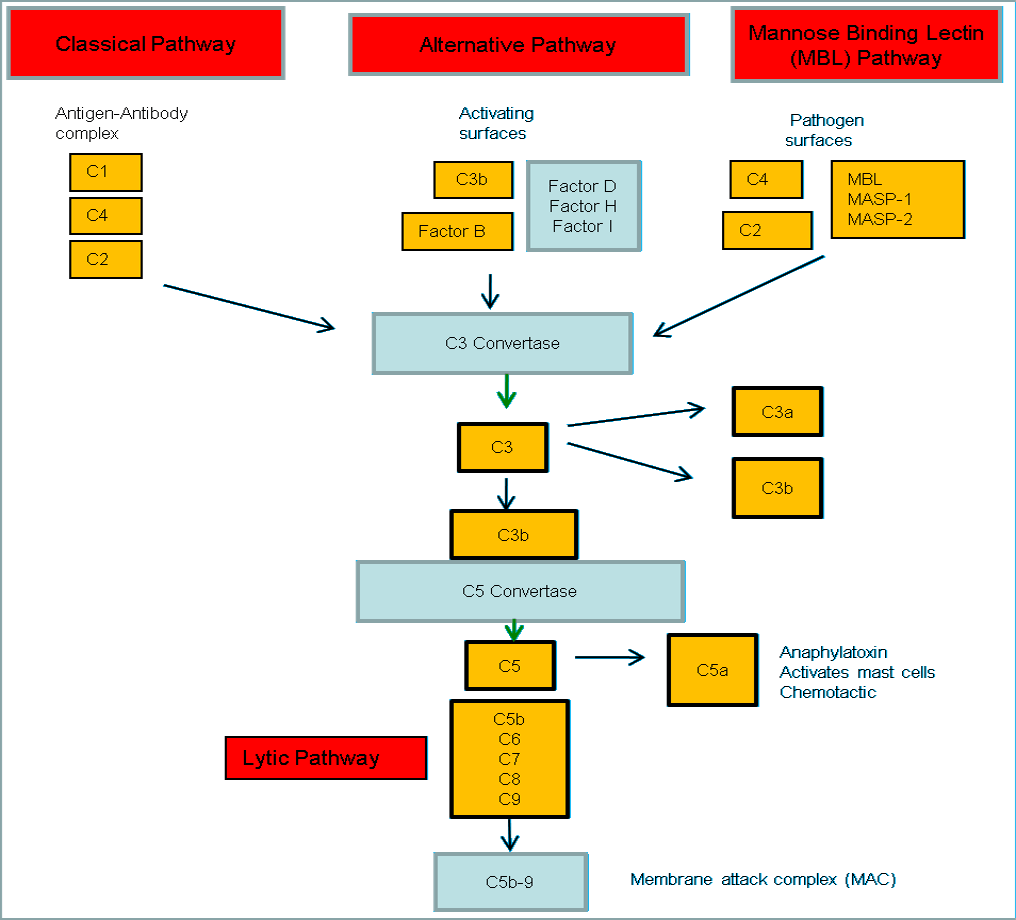
The complement cascade can be activated in 3 ways:
- Classical pathway
- Alternative pathway
- Lectin pathway
C3 and C3 CONVERTASE
All of the 3 pathways converge at a particular point in the cascade and produces a common complex called C3-convertase which cleaves the C3 component of the complement system.
C5 and MEMBRANE ATTACK PATHWAY
No matter, how the complement system is activated, generation of C5 convertase is important for the culmination of the complement mediated effector mechanisms, leading to the formation of Membrane Attack Complex (MAC).
THE ALTERNATIVE PATHWAY
It is phylogenetically more primitive pathway compared to the classical pathway but was named “alternative” probably because it was discovered later among the two. This pathway consists of proteins known by the term “Factors” like Factor B, Factor D.
Steps:
- Spontaneous hydrolysis of C3.
- C3b product is very reactive and can bind to invader’s cell surface.
- If C3b cannot find the cell surface to bind within 60 microseconds – it is hydrolyzed.
- Factor B binds C3b on cell surface.
- Factor D cleaves Factor B, leaving “Bb” which has a “chainsaw mechanism”
- C3bBb acts as C3 convertase which cleaves C3 – this creates a continuous loop and many C3b can be deposited on the invader’s cell surface.
- C3bBb together with another C3b molecule can act as C5 convertase which cleaves C5
- C5b together with C6, C7, C8 and C9 forms MAC
- C5b, C6, C7, and C8 form a “stalk” that anchors the complex in the bacterial cell membrane. Then C9 proteins are added to make a channel that opens up a hole in the surface of the bacterium.
THE CLASSICAL PATHWAY
The classical pathway is phylogenetically newer and is named “classical”, only because it was discovered earlier. It is activated by Antigen-Antibody complexes. The classical pathway consists of 4 proteins C1, C2, C3 and C4.
Steps:
- C1 is a complex made of hexamer C1q and serine proteases C1r and C1s.
- C1q binds to Fc region of antibody. IgM is more efficient because of it’s high avidity as it can bind to 3 C1q compared to IgG which can bind to only one.
- Activated C1s cleaves C4 and C4b either binds to antigen-antibody complex or to the adjacent cell surface.
- C1s also cleaves C2 and the C4bC2a complex is the C3 convertase of classical pathway.
- Once C3b is formed – it can either follow alternative pathway or bind to C4bC2a (C3 convertase) to form C5 convertase.
- Similarly, MAC is formed.
C1 binds to Fc portion of IgM or IgG near hinge-region. This region is exposed when the antigen binds to the antibody.
THE LECTIN PATHWAY
The lectin pathway consists of C2, C3, C4 and some calcium-dependent lectin family proteins which are homologous to C1 component.
Steps:
- In the blood, MBL (Mannose Binding Lectin) binds to another protein called MASP (Mannan Associated Serine Protease).
- When MBL grabs its target (mannose on the surface of a bacterium, for example), the MASP protein functions like a convertase to clip C3 complement proteins to make C3b.
Mnemonic for Complement system:
- Classical pathway: Complex (Immune complex) activated; C1 + C4 – C2 = C3
- Mannose binding lectin (MBL) pathway: Microbe mannose activated
- Alternative pathway: Alien particles (like microbe or tumor cells) activated; Factors mediated
OTHER FUNCTIONS OF COMPLEMENT
1. C3a, C4a, C5a:
- Anaphylotoxins; Chemoattractants
- C5a is the most powerful
2. iC3b:
- Inactive form of C3b cleaved by serum proteins
- Oposonin
PROTECTION OF NORMAL CELLS FROM COMPLEMENT
- Membrane Co-factor Protein (MCP or CD46) accelerates clipping of C3b. CD46, Factor H and Factor I prevent excessive alternate pathway activation.
- Decay Accelerating Factor (DAF) accelerates destruction of C3 convertase (C3bBb).
- Cell surface protein CD59 (protectin) prevents incorporation of C9 into nascent MACs (i.e. inhibits formation of MAC).
COMPLEMENT DEFICIENCIES
Even though gram-positive bacteria may be resistant to the membrane attack complex (MAC), the early components of the cascade mediate localized inflammation and opsonize the bacteria.
Most common complement deficiecy is that of C2 which manifests as recurrent pyogenic infections and SLE like syndrome in childhood.
| Complement Deficiency | Disease |
| CD46, factors H and I | Atypical or Non-epidemic Hemolytic Uremic Syndrome (HUS) |
| C5-C8
C3b and C3b inactivator | Severe Neisseria and Toxoplasma infection infections
Recurrent pyogenic infection |
| Early complements C1, C4, C2. | Systemic lupus erythematosus (SLE), glomerulonephritis and polymyositis (increased immune complexes) |
| C1-inhibitor | Hereditary angioedema (uncontrolled C1 activation) |
| DAF and CD59 | Paroxysmal Nocturnal Hemoglobinuria (PNH) |
Eculizumab prevents the conversion of C5 to C5a. This inhibitor not only reduces the hemolysis and attendant transfusion requirements in patients of paroxysmal nocturnal hemoglobinuria (PNH), but also lowers the risk of thrombosis.
References:
1. A Textbook of Immunology – J.K Sinha and S.Bhattacharya
2. How the Immune System Works – Lauren Sompyrac
3. Complement System – British Society of Immunology
4. Complement Cascade – Studentreader
5. Kaplan USMLE Step 1 – Immunology and Microbiology

He is the section editor of Orthopedics in Epomedicine. He searches for and share simpler ways to make complicated medical topics simple. He also loves writing poetry, listening and playing music. He is currently pursuing Fellowship in Hip, Pelvi-acetabulum and Arthroplasty at B&B Hospital.

very elaborate.
concise and helpful!