Vmax is the maximum velocity, or how fast the enzyme can go at full ‘‘speed.’’ Vmax is reached when all of the enzyme is in the enzyme–substrate complex.
- Vmax is directly proportional to the enzyme concentration.
Km is the substrate concentration at which v = 1/2 Vmax. Km approximately describes the affinity of the substrate for the enzyme. The lower the value of Km, the higher the apparent affinity for substrate.
The closer to 0 on the Y-axis, the higher the Vmax
The closer to 0 on the X-axis, the higher the Km.
Mnemonic:
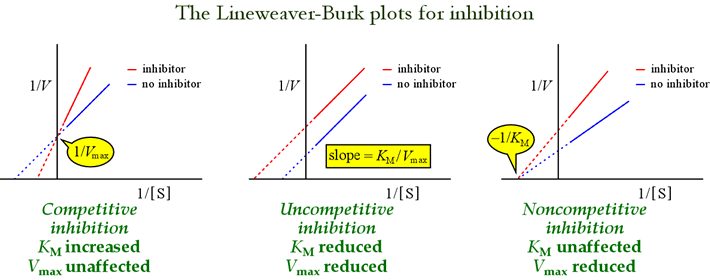
- Competitive inhibitor (Km-pitive inhibitor): Km increases, Vmax doesn’t change
- Non-competitive inhibitor (Non-Km-pitivie inhibitor): Km doesn’t change, Vmax decreases
Competitive inhibition:
These are structurally similar to substrates and hence competes with substrate to bind at active site of enzyme (cannot bind to enzyme substrate complex). Increasing the substrate can overcome inhibition as overall efficacy of enzyme is not affected (more substrate is needed to achieve 1/2 Vmax, i.e. Km increases).
Non-competitive inhibition:
These are structurally different from substrates and hence bind enzymes at sites distinct from substrate binding site and reduce the enzyme activity (i.e. no competition with substrate). It can bind to both the enzyme and enzyme-substrate complex. Increasing the substrate will not overcome the inhibition, hence, Vmax decreases and hence, Km remains same.
- May be irreversible or reversible
Uncompetitive inhibitors:
These are like non-competitive inhibitiors but, they only bind to the enzyme when substrate is bound to the enzyme (i.e. binds to enzyme substrate complex only). Uncompetitive inhibitors decrease both Vmax and Km.
Graph:
Mnemonic:
- Vmax = Vertical (y-axis)
- Km = x-axis (‘k’ looks like ‘x’)
- Very efficient and Com(Km)petent, i.e. Vmax represent efficacy and Km represent competency.
Competitive inhibitors:
- Competitive inhibitors Cross at y-axis (i.e. Vmax is same).
Non-competitibe inhibitors:
- Doesn’t cross but converge at x-axis (i.e. Km is same).
Uncompetitive inhibitors:
- Follow separate path to the left (both Vmax and Km is decreased).
| Competitive inhibitors, reversible | Competitive inhibitors, irreversible | Noncompetitive inhibitors | |
| Resemble substrate | Yes | Yes | No |
| Overcome by increasing [S] | Yes | No | No |
| Bind active site | Yes | Yes | No |
| Effect on Vmax | Unchanged | Decreased | Decreased |
| Effect on Km | Increased | Unchanged | Unchanged |
| Pharmacodynamics | Decreased potency | Decreased efficacy | Decreased efficacy |

This was very helpful, it really helped me with my bioprocess engineering lecture. Thank you very much 🙂