Liver as a hub of cholesterol metabolism
- All nucleated cells have the capacity to synthesize cholesterol from acetyl-CoA (like most biological lipids).
- 90% of synthesis occurs in liver and intestine.
- The cholesterol biosynthesis pathway involves enzymes that are in the cytoplasm, microsomes (ER), and peroxisomes.
Liver obtains cholesterol from 3 major sources:
- Chylomicron remnants (dietary cholesterol)
- Denovo cholesterol synthesis
- LDL (endogenous cholesterol)
Liver uses cholesterol in 2 major ways:
- Synthesize bile acids and also includes cholesterol and cholesteryl esters in the bile.
- Exports cholesterol in the blood in the form of VLDLs.
Cholesterol esterification:
Free cholesterol can be taken up by lipoproteins, but is confined to the outer surface of the particle. By converting cholesterol to cholesteryl esters more cholesterol can be packaged into the interior of lipoproteins. This vastly increases the capacity of lipoproteins, allowing for more efficient cholesterol transport through the blood stream. Esterification is catalyzed by 2 enzymes:
- In tissues: ACAT (Acyl: cholesterol acyl transferase) – upregulated by cholesterol
- In plasma: LCAT (Lecithin acyl transferase) – upregulated by Apo-A1 in HDL
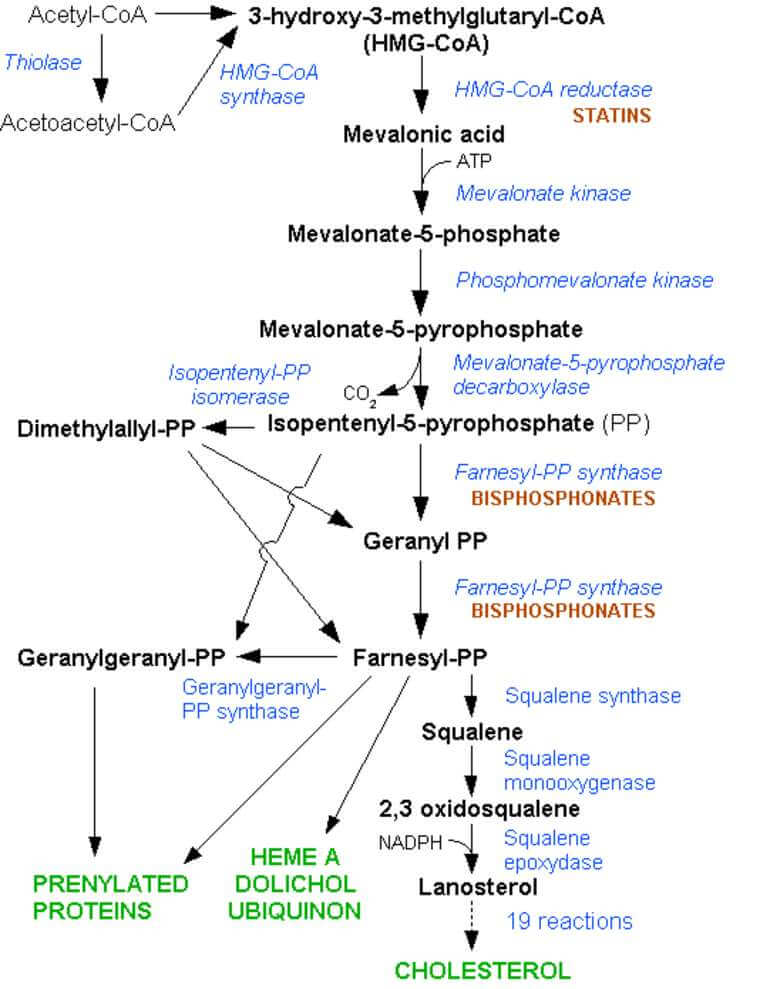
Cholesterol synthesis
Mnemonic
“Ah Ah Help Me – Plan In Diet Good Food- Stay Low in Cholesterol”
Correlate with the mnemonic above:
- Acetyl-CoA (Ah!) – 2C
- Acetoacetyl-CoA (Ah!) – 4C
- HMG CoA (Help) – 6C
- Mevalonate (Me) – 6C
- Phospho-mevalonate and Pyrophospho-mevalonate (Plan) – 6C
- Isopentanyl pyrophosphate (In) – 5C
- Dimethylallyl pyropphosphate (Diet) – 5C
- Geranyl pyrophosphate (Good) – 10 C
- Farnesyl pyrophosphate (Food) – 15C
- Squalene (Stay) – 30C
- Lanosterol (Low in) – 30C
- Cholesterol (Cholesterol) – 27C
Some important steps to remember:
1. Acetyl-CoA is shifted from mitochondria to cytoplasm by citrate shuttle.
2. HMG-CoA reductase converting HMG-CoA to Mevalonate is the rate limiting enzyme.
- 2 NADPH are used (generated from HMP pathway)
- Inhibited by:
- Feedback inhibition: Mevalonate, Cholesterol, Bile acids
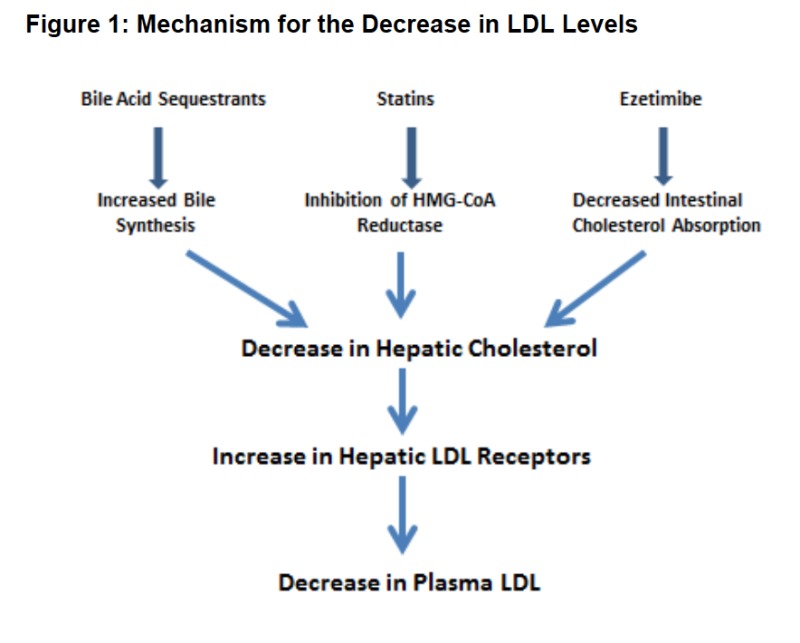
- Reversible competitive inhibition: Statins (decreased cholesterol synthesis leads to increased LDL receptors leading to internalization of cirulating lipoproteins)
- Covalent modification (phosphorylation): Glucagon
- Activated by: Insulin (dephosphorylation), Thyroid hormone
Bile acid sequestrants (Cholestyramine, Cholestipol) reduce bile acid absorption – hence, increasing hepatic conversion of cholesterol to bile acids and also increase LDL receptor. Ezetimbie (intestinal cholesterol absorption inhibitior) also increases LDL receptor by same mechanism.
PCSK9 Inhibitors (alirocumab and evolocumab) are monoclonal antibodies that bind to proprotein convertase subtilisin/kexin type 9 (PCSK9). PCSK9 destroys LDL-receptor on hepatocytes, which results in decreases clearance of LDL.
Inhibition of PCSK9 improves clearance of LDL.Fibrates (gemfibrozil, bezafibrate, and fenofibrate) activates PPAR-α:
- upregulates lipoprotein lipase (reduces levels of triglycerides) – by increased clearance of TG rich chylomicrons and VLDL
- induces HDL synthesis
- downregulates cholesterol-alpha-7-hydroxylase (decreased conversion of cholesterol to bile – risk of gallstones)
Niacin (Vitamin B3) inhibits hormone-sensitive lipase (decreased free fatty acid mobilization and decreased TG synthesis) thus inhibiting hepatic VLDL synthesis and thus LDL formation. It also decreases HDL catabolism receptor, thus decreasing HDL uptake and removal. Niacin (nicotinic acid) decreases renal urate excretion by competing with the same transporter in proximal renal tubule and cause hyperuricemia.
3. Mevalonate (6C) is sequentially phosphorylated using ATP by 3 kinases (3 ATP used) and then decarbocylated to form Isopentenyl pyrophosphate (5C). Isopentenyl pyrophosphate can also go into synthesis of: Vitamine A, E and K.
4. 6 molecules of isoprenoid units (5 C) condenses to form Squalene (30 C). Conversion of Farnesyl PP (15 C) to Squalene (30 C) is catalyzed by Squalene synthase (Farnesyl PP Farnesyl transferase) which requires 2 NADPH. Franesyl PP is important for:
- Synthesis of CoQ (Electron Transport Chain)
- Synthesis of Heme-a protein (Prosthetic group for cytochrome c oxidase)
- Synthesis of Dolichol PPi for N-linked glycosylation of proteins
5. Squalene is cyclized to lanosterol (resembles steroid nucleus) using 2 enzymes, one of which is squalene epoxidase (1 NADPH used).
6. With series of reactions, squalene forms desmosterol (24-dehydrocholesterol) in ER membrane. Desmosterol reductase (inhibited by Triparanol) converts desmosterol into cholesterol.
Mechanism of Rhabdomyolysis by Statins
Statins block production of Farnesyl PPi by blocking HMG CoA reductase in the synthesis of lipid pathway. Farnesyl PPi is needed for production of CoQ. Decreased Farnesyl PPi leads to decreased CoQ required in ETC. Decreased CoQ results in Decreased ATP production in muscles results in dysfunctioning of Sodium/K pump leads to cell swelling and rhabdomyolysis.
Nitrogen Containing Bisphosphonates
Nitrogen containing bisphosphonates like Zoledronate and Alderonate inhibits Farnesyl PPi synthase and Farnesyle PPi is needed for osteoclast survival.
Smith‐Lemli‐Opitz syndrome (SLOS)
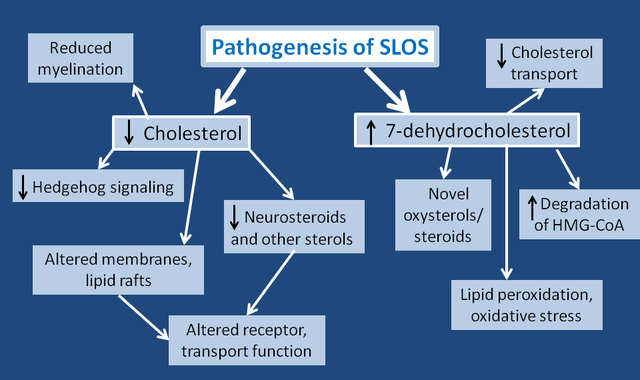
- Most common and best understood of the inborn errors of cholesterol metabolism
- Caused by mutations in DHCR7 which encodes the final enzyme in the cholesterol synthetic pathway
- Cholesterol is necessary for normal embryonic development and has important functions both before and after birth. It is a structural component of cell membranes and the protective substance covering nerve cells (myelin)
- Clinical features due to cholesterol deficiency and/or accumulation of cholesterol precursors and their metabolites:
- Autism
- Malformation of heart, lungs, kidney, gastrointestinal tract, genitalia
- Hypotonia
- Syndactyly and Polydactyly
- Supplementation of cholesterol is a commonly employed potential therapy

He is the section editor of Orthopedics in Epomedicine. He searches for and share simpler ways to make complicated medical topics simple. He also loves writing poetry, listening and playing music.
Thank you! Epomedicine..
Your mnemonics are a blessing