This article is in response to the request made by a reader in the comment section. Today, we will try to simplify the biliary tree cancers.
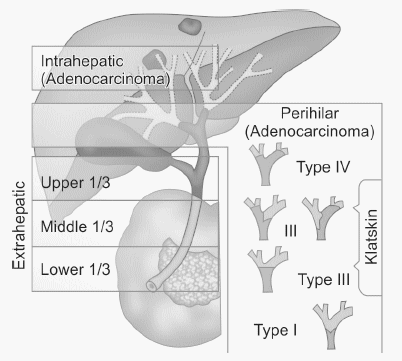
Classification of Cholangiocarcinoma
Second-order bile ducts act as the separation point between intrahepatic and extrahepatic cholangiocarcinoma.
A. Inhtrahepatic – 10 to 20%
B. Extrahepatic
- Hilar bile duct cancer (Klatskin tumor) -60% (most common location is proximal 1/3rd)
- Distal bile duct cancer – 20 to 30%1
Risk factors for Cholangiocarcinoma
Remember the 14 “C”s of Cholangiocarcinoma.
- Classic people: >50 years
- Close to China (Indochina) distribution: Cambodia, Vietnam, Laos, Thailand
- Clonorchis sinensis and Opisthorchis viverrini (Liver flukes)
- Chronic typhoid carriers
- Choledochal cyst
- Caroli’s disease (Todani classification type V)
- Colitis (Ulcerative colitis)
- Cholangitis (Primary sclerosing cholangitis)
- Primary sclerosing cholangitis is the commonest etiology in Western world.
- Early cholangiocarcinoma at the age of 30-50 years.
- Contrast material: Thorotrast
- Contraceptive pills
- C type hepatitis
- Cirrhosis
- Chemical carcinogen: Vinyl chloride, Dioxins, Asbestos
- Connection: Biliary-enteric anastomosis
p53 mutations are common in intrahepatic cholangiocarcinoma.
K-ras mutations are common in extrahepatic cholangiocarcinoma. [footnoteDiagnostic Histopathology of Tumors By Christopher D. M. Fletche[/footnote]
Histology of Cholangiocarcinoma
- Almost all are: Adenocarcinoma
- Others: Squamous cell carcinoma, Sarcoma
- Most common: Sclerosing adenocarcinoma
- Most favorable prognosis: Papillary adenocarcinoma
Modified Bismuth-Corlette classification for Perihilar Cholangiocarcinoma
Type I: Common hepatic duct
Type II: Confluence without involvement of secondary ducts
Type IIIa: Type II + Right secondary intrahepatic ducts
Type IIIb: Type II + Left secondary intrahepatic ducts
Type IV: Secondary ducts on both sides
Clinical features of Cholangiocarcinoma
| Features | Intrahepatic cholangiocarcinoma | Perihilar cholangiocarcinoma | Distal cholangiocarcinoma |
| Non-specific signs and symptoms (Weight loss, pain, anorexia, ascites) | Present | Present | Present |
| Peripheral liver mass | 1/3rd cases (but hepatic bruit is absent which may be present in Hepatocellular carcinoma) | – | – |
| Obstructive jaundice onset – pruritus, icterus, dark urine, pale stool | + (Uncommon but more frequent than in Hepatocellular carcinoma) | ++ (Early if confluence of hepatic ducts involved that right or left alone) | +++ (Early presentation) |
| Courvoisier’s sign (palpable gallbladder) | – | – | + (Obstruction distal to cystic duct) |
- Assess to identify the underlying risk factors mentioned above as 14 “C”s.
- Patient may present with features of cholangitis in extrahepatic cholangiocarcinoma.
- Intermittent jaundice can occur as a result of:
- Intermittent shedding of papillary tumors
- Ball-valve action of mobile growth
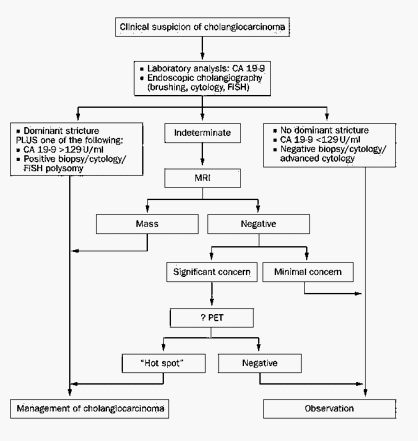
Diagnostic criteria for Cholangiocarcinoma
1. Malignant appearing stricture AND persisting serum CA 19-9 level >129 U/ml in the absence of bacterial cholangitis
2. Mass lesion on cross-sectional imaging
3. Positive conventional cytology result
4. Positive (transluminal) biopsy specimen
5. Stricture plus polysomy on Fluorescence in-situ hybridization (FISH) 2
Investigations for Cholangiocarcinoma
A. Liver Function Tests (LFT):
- Suggesting obstructive jaundice (Cholestasis): Increased conjugated bilirubin, ALP, GGT, Raised PT/INR
- Suggesting hepatic injury: Raised aminotransferases (more common in intrahepatic cholangiocarcinoma with direct hepatic invasion)
B. Tumor markers: There are no specific tumor markers for cholangiocarcinoma but the commonly used in diagnosis are:
- Serum CA 19-9 (value >100 ng/ml in absence of bacterial cholangitis strongly suggest cholangiocarcinoma)
- Serum CEA
- Serum CA-125
Tumors stain positively for Cytokeratin 7, 8 and 19 and negatively for cytokeratin 20.
C. To identify tumor location (mass or level of biliary obstruction) and characterize extent of involvement around portal vein and hepatic artery:
- Ultrasonograpgy (USG) – 1st line
- Contrast enhanced CT (CECT) or MRI (more sensitive)
- MR angiography (staging investigation)
D. To define extent of disease in secondary hepatic ducts, obtain brushings for cytology and for biliary decompression with stents:
- Proximal bile duct obstruction: PTC (Percutaneous transhepatic cholangiography) is preferred
- Distal bile duct obstruction: ERCP (Endoscopic retrograde Cholangio-pancreatography) is preferred
- MRCP (Magnetic Resonance Cholangio-pancreatography):
- Advantage: Non-invasive and it can assess the extent of duct involvement above and below the level of obstruction.
- Disadvantage: Only diagnostic value (biliary decompression cannot be preformed like in PTC or ERCP)
Yield of brushing cytology alone: Only 30% (because of desmoplastic reaction surrounding the bile duct and extending into submucosal space).
Yield of brushing cytology with biopsy: 40-60%
TNM and Staging of Cholangiocarcinoma
Discussing the TNM and staging of all types of cholangiocarcinoma is beyond the scope of this article. Here, we will discuss only about the hilar cholangiocarcinoma. Just remember that TNM for all 3 types of cholangiocarcinoma are different.
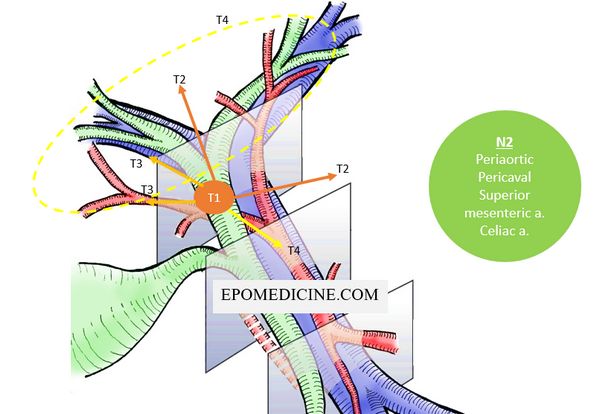
Stage IV disease:
- Distant metastasis (M1) or
- Nodal metastasis (N2) to major artery/vein lymph nodes (periaortic, pericaval, superior mesenteric artery, and celiac artery lymph nodes) or
- Tumor invades (T4):
- Vein: Main portal vein or bilateral branches Or
- Artery: Common hepatic artery Or
- Biliary tract: Bilateral 2nd order biliary radicals Or
- Biliary tract + Vein or Artery: Unilateral 2nd order biliary radicals + Contralateral portal vein or hepatic artery
Stage III disease:
- Tumor invades unilateral branches of the portal vein or hepatic artery (T3) Or
- Regional lymph node (N1) involvement (nodes along the cystic duct, common bile duct, hepatic artery, and portal vein)
Tumor confined to Bile duct wall (T1) without nodal involvement (N0) or metastases (M0): Stage I disease
Rest fall into stage II disease.
Contraindications to Resection with Curative Intent
| A. | Patient Factors |
| |
| B. | Local Factors |
| |
| C. | Distant Disease |
|
Surgical therapy for Cholangiocarcinoma
1. Intrahepatic cholangiocarcinoma: Similar to Hepatocellular carcinoma
- Segmentectomy or Lobectomy to achieve R0 resection based on tumor size.
- Pre-operative portal vein embolization and biliary drainage may reduce morbidity and mortality.
2. Perihilar cholangiocarcinoma:
a. Bismuth I and II: Local resection of tumor and adjacent nodes.
b. Bismith IIIa or IIIb: Resection of involved hepatic lobe may also be planned.
Bismuth IVb is not resectable.
3. Distal cholangiocarcinoma:
a. Proximal 1/3rd: Resection of tumor + Hepaticojejunostomy
b. Middle 1/3rd: Resection of tumor + End-to-end bile duct anastomosis (if possible) or Hepaticojejunostomy
c. Distal 1/3rd: Pacreatico-duodenectomy (Whipple’s procedure)
Liver transplant in Cholangiocarcinoma in case of Non-resectability
- Locally advanced disease (typically hilar) involving the surrounding large vessels (portal vein, hepatic artery) and extension to secondary biliary radicles.
- Primary sclerosing cholangitis
- Hepati dysfunction
Palliative Chemotherapy ± Radiotherapy in Cholangiocarcinoma
Role of neoadjuvant chemotherapy in cholangiocarcinoma has not been established.
5-FU, Gemcitabine or Capecitabine can be used with radiotherapy as adjuvant chemoterhapy.
Combination of:
- Gemcitabine
- Platinum compounds
Other palliative measures
1. Photodynamic therapy (PDT): A photosensitizer is administered and selectively retained by the target tumor tissue. The photosensitizer is nontoxic in its native state, however, after activation by a light at a particular wavelength, the photosensitizer becomes cytotoxic and produces local tissue destruction. The only relevant side effect seen to date is phototoxicity, which lasts often for 4-6 week.
2. Radiofrequency ablation: RFA may provide successful local tumour control in patients with intermediate (3-5m) or small (<3cm) intrahepatic nodules.
3. Transarterial chemo-embolisation (TACE): It reduces oxygen and nutrients to tumor and increases the local drug concentration and reduces drug clearance from liver. Chemoembolisation with drug-eluting beads combines the drug with the embolisation device by using the embolic device to reduce blood flow to the tumour whilst at the same time eluting a chemoherapeutic agent into the tumour via its own vasculature.
4. Transarterial Radioembolisation (TARE) with yttrium 90: Permanent DNA damage is caused by one or both DNA strands, and apoptosis is initiated or reproductive death is eventually achieved.
Prognosis of Cholangiocarcinoma
Following resection, the median survival is approximately 18 months, with 20 per cent of patients surviving five years postresection.
Survival appears better for distal tumours compared to those involving the upper third of the biliary tree.
Although gallstones are considered as predisposing factors for carcinoma gallbladder, CBD stones are not considered risk factors for cholangiocarcinoma.