A 12 year old female patient came to OPD with the chief complaints of increased thirst and increased frequency of urination for last 1 year. She drank about 10-11 liters of water everyday and passed similar amount of urine with a frequency of 10-12 episodes on daytime and 3-4 episode on night time. This had been going on for the last 1 year with no improvements despite many hospital visits. Due to frequent urination, she was less attentive in classes and her academic performance was deteriorating according to her teacher.
There was no history of:
- Urgency, Hesitancy, Burning micturition (UTI)
- Bedwetting during day or night time (Eneuresis and Polyuria)
- Head injury (Central Diabetes Insipidus)
- Fever, headache or vomiting, blurred vision, photophobia and diplopia (Intracranial Space Occupying Lesion/Meningitis)
- Abnormal body movement or abnormal behavior
- Drug intake for chronic period
- Bone pain
- Cough
- Weakness of any body parts
There was a history of headache about 1 and 1/2 year back which was gradual in onset and generalized but non progressive and was evaluated in hospital. She was found to have papilledema the cause of which could not be ascertained and she was better with symptomatic medication.
Historical diagnosis:
- Primary polydipsia
- Diabetes insipidus
- Diabetes mellitus
- General condition: Conscious, Oriented to time, place and person
- No pallor, icterus, cyanosis, edema, dehydration
- Vital signs: Temperature – 98.2, RR – 20/min, Pulse – 72/min, BP – 100/70 mmHg
- Anthropometry: Weight – 29.5 kg (5th percentile), Height – 139 cm (10th percentile)
CNS examination:
- Conscious, oriented and cooperative.
- Intelligence and memory intact. Speech normal.
- Vision normal, no nystagmus.
- Bilateral pupil round regular and reactive. Eye movement and field of vision normal.
- Fundus examination: bilateral disc edema.
- Other cranial nerves were intact.
- Bulk, tone, power were normal of all major muscle groups.
- Deep tendon and superficial reflexes were normal.
- No sensory or motor deficit.
Other systemic examinations: Grossly normal
Provisional diagnosis:
- Central DI
- Primary polydipsia
- Nephrogenic DI
- Diabetes mellitus
Plan of action:
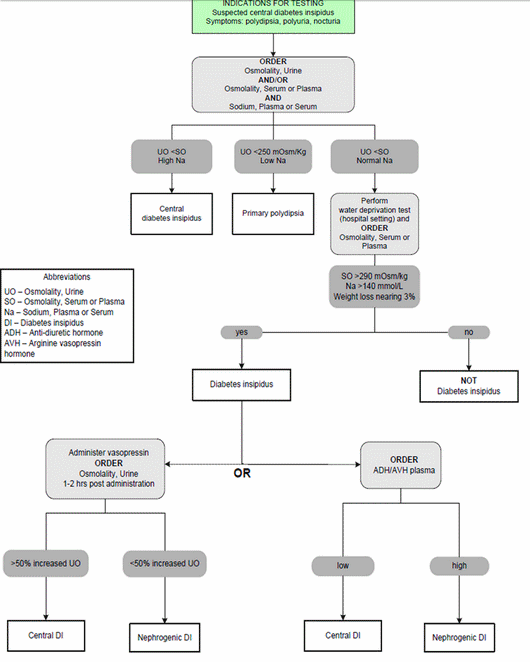
- Confirmation of polyuria and polydipsia
- Baseline electrolytes, counts and urine examination.
- Measurement of serum and urine osmolality during ad libitum food and water intake.
- Perform water deprivation test.
- Imaging for central cause.
Initial lab parameters:
| Parameters | Values |
| Polyurea | 180ml/kg/day |
| Polydipsia | 6 litres of water in 24 hour |
| Na/K/CaGlucose | 137 meq/lt, 4.2 meq/lt, 2.2 mmol/lt3.9 mmol/lt |
| Urea/creatinine | 3.2mmol/lt, 63micromol/lt |
| Serum osmolality | 287.9 mosm/kg/water |
| Urine osmolality | 76 mosm/kg/water |
| CBC | Hb- 12gm%, TC- 6700/cmm,Platelets- 2,29,000/cmm |
| Urine RME | Sugar-nil. Albumin-nil. Pus cells 0-2/hpfSpecific gravity- 1.003. |
Water deprivation test:
| TIME | WEIGHT | URINE SP. GR. | URINE OSM. | SERUM Na | SERUM OSM. | URINEOUTPUT |
| 9AM | 27.7kg | 1.002 | 76 | 137 | 287 | 550ml |
| 12PM | 26.9kg | 1.003 | 112.8 | 142 | 299.5 | 450ml |
| 3PM | 26.8kg | 1.005 | 164.4 | 142 | 299.9 | 250ml |
| Desmopressin | Administered | intranasally | ||||
| 5PM | 26.5kg | 1.009 | 255.7 | 143 | 301.8 | 25ml |
MRI Head: Normal findings
Final Diagnosis:
- Idiopathic Central Diabetes Insipidus
Treatment:
Intranasal Desmopressin 10 μg twice daily
[hr]
DIABETES INSIPIDUS
A) Definitions:
Polyuria is defined by a urine volume in excess of 2 l/m2/24 hr
| Age | Amount |
| At birth and neonate | 150 ml/kg/24 h |
| Untill 2 years of age | 100–110 ml/ kg/24 h |
| Older child | 40–50 ml/kg/24 h |
Polydipsia is simply defined:
- by excessive drinking based on nurses estimates. (Jose et al, BJPSYCH, 1991, polydipsia in psychiatric illness)
- as drinking of water >4 lt/day (Blum, 1983)
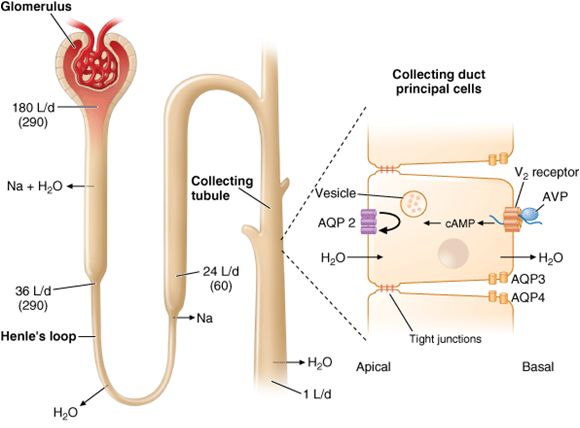
Diabetes insipidus is a disease in which large volumes of dilute urine (polyuria) are excreted due to
- Vasopressin (AVP) deficiency
- AVP resistance
- Excessive water intake
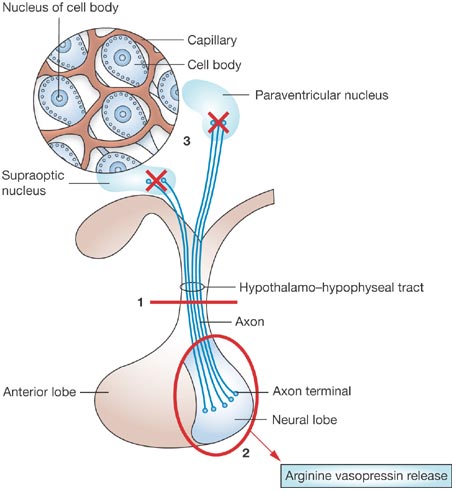
Central diabetes insipidus (CDI) is the end result of a number of conditions that affect the hypothalamic-neurohypophyseal system.
B) Pathophysiology:
C) Epidemiology:
National Surveillance of Central Diabetes Insipidus (CDI) in Denmark – Results from 5-years Registration of 9,309 Prescriptions of Desmopressin to 1,285 CDI patients.
- Period prevalence rate: 23 CDI patients per 100,000 inhabitants, with higher prevalence in children and older adults above 80 years of age.
- Yearly incidence rate of new cases: 3-4 per 100,000.
- Incidence of (presumable) congential CDI: 2 per 100,000 infants.
D) Etiology:
Congenital Central DI
| DEFECT | TYPES |
| GENE DEFECT |
1.AVP-NP II GENE DEFECT
Autosomal dominant type Autosomal recessive type 2. WFS 1 GENE DEFECT ( DIDMOAD SYNDROMME) |
| STRUCTURAL DEFECT |
1.EMPTY SELLA SYNDROMME
2.SEPTO-OPTIC DYSPLASIA
3.STALK DEFECT
4.AGENESIS OF CORPUS CALLOSUM
5.RATHKE CLEFT DEFECT
6.VASCULAR MALFORMATION
|
Acquired Central DI
| CAUSES | INCLUSIONS | |
| TUMOURS | GERMINOMA CRANIOPHARYNGIOMA GLIOMA |
PINEALOMA HEMANGIOMA METASTASIS |
| INFILTRATIVE LESIONS | TUBERCULOSIS SARCOIDOSIS HISTOCYTOSIS |
HEMOCHROMATOSIS AMYLOIDOSIS |
| DRUGS | PHENYTOIN CARBAMAZEPINE |
VALPROIC ACID ADRENALINIC AGONISTS |
| TRAUMA | HEAD INJURY | CRANIAL SURGERIES |
| CEREBRAL INSULT | HYPOXIC ENCEPHALOPATHY |
E) Clinical features:
Although the signs and symptoms of DI vary with the etiology, it manifests differently in different age groups depending on their ability to replenish water.
| Age | Presentation |
| Infants | vigorous suck, vomiting, recurrent episodes of fever without an apparent cause, excessive crying, irritability, weight loss, constipation, and excessively wet diapers. |
| Younger children | primary enuresis and difficulty in toilet training. |
| Older children | characteristically have high urine output and nocturia leading to disturbed sleep and easy fatigability. |
F) Laboratory diagnosis:
- The initial step in the diagnosis of DI is to ascertain the presence of polyuria which can be established with an accurate 24 hours urine output measurement
- Urine output more than 4 ml/kg/hr in infants and children and more than 6 ml/kg/hr in newborn is suggestive of polyuria.
- Once polyuria is established, it is necessary to rule out solute diuresis i.e. glycosuria, hypercalciuria or uremia by urine analysis and biochemistry.
- Measurement of serum potassium and calcium concentrations is also important to exclude the possibility of polyuria secondary to hypokalemia or hypercalcemia.
- Presence of polyuria in the absence of solute diuresis should raise the suspicion of DI.
- Urine osmolality of more than 800 mOsm /kg with a serum osmolality of less than 270 mOsm/kg rules out the diagnosis of DI.
- Dilute urine with an osmolality of less than 300 mOsm/kg, and a serum osmolality of more than 300 mOsm/kg effectively establishes the diagnosis
- If the initial serum osmolality is less than 300 mOsm/kg, a water deprivation test is done to distinguish between CDI and NDI.
- Various studies suggest that the combination of the water deprivation test and direct AVP determination would allow the diagnosis of more than 95% of all cases of DI correctly.
Water deprivation test:
- This is a potentially hazardous test and close medical supervision is required.
- The test must be arranged in advance with the Chemical Pathology Laboratory and all specimens handled urgently.
- Consider overnight admission prior to test..
- A light breakfast with minimal fluid (water) may be taken before the test starts.
- Weigh the patient accurately, to within 0.1 kg. Use the same scales throughout the test.
- Start test. Withhold fluids for up to 7 hours, with constant supervision. Empty bladder; measure urine osmolality and sodium concentration.
Pitfalls of water deprivation test
- The dehydration test has been questioned as the diagnostic reference standard for several reasons.
- The long dehydration period may provide not only an osmotic but also a volume stimulus
- It has been shown that dehydration alone may induce considerable AVP-independent urine concentration.
- Chronic polyuria per se impairs the renal medullary concentrating gradient and down-regulates aquaporin-2 synthesis, and hence response to desmopressin can be attenuated in patients with DI.
Direct AVP measurement:
- It is required that whenever the direct test is applied, the osmotic stimulus has to be pursued until hypertonic levels of serum osmolality have been attained (>295 mOsm/kg).
- AVP assay has failed to be a diagnostic reference standard to date due to its methodological limitations- a very short half life of 10 -30 minutes, high pre analytical instability and high turn around time in most laboratories.
Measurement of Aquaporin-2 excretion:
- The urinary excretion of Aquaporin-2 increases following desmopressin administration in CDI, while there is no increase in NDI.
- Plasma copeptin levels
- More recently plasma copeptin levels have been studied as a surrogate marker of AVP.
- Copeptin, the C-terminal part of the AVP precursor, co-secreted with AVP is much easier to measure.
- Plasma copeptin is a valuable surrogate of AVP release in patients with the DI, exhibiting a promising diagnostic potential.
MRI Pituitary:
MRI pituitary is an important tool for assessment of etiology of CDI, and should always be performed after gadolinium injection, to check for abnormal enhancement within the stalk. It is also important to perform MRI after the patient is well hydrated as the intensity of posterior pituitary bright signal is inversely related to the degree of dehydration. MRI findings have been heterogenous:
- Absence of posterior pituitary bright signal: In most of the patients with CDI the hyperintense signal of the posterior pituitary was absent at diagnosis; however its presence does not rule out a diagnosis of CDI.
- The size of the adenohypophysis
- Pituitary stalk thickness (PST): The pituitary stalk is considered enlarged if at least part of the stalk is found to have a diameter of > 2.0 mm. PST is observed in almost one third of children with CDI, and may be the first sign of a germinoma (15%) or of stalk infiltration, as in Langerhans cell histiocytosis (LCH – 15%). CDI with PST remains idiopathic (70%) in most cases. In idiopathic PST, repeat MRI and tumor marker estimation is done every 3 – 6 months during the first 3 years. Subsequently, MRI evaluation is performed once per year for 2 years and every 2 – 5 years thereafter, depending on the size and progression of the lesion.
Other Causes:
Gene analysis: Genetic defects in the synthesis of vasopressin are typically inherited as an autosomal dominant trait and caused by mutations in the AVP-NPII gene, but autosomal recessive defects have also been described. More than 50 mutations in the AVP-NPII gene have been reported. Genomic DNA can be analysed for AVP gene mutations using polymerase chain reaction (PCR) amplification and sequencing.
Autoimmunity: Circulating vasopressin-cell autoantibodies (AVPc- Abs) have been detected in 23%-75% of young patients with CDI, indicating that autoimmune CDI might account for a significant number of the idiopathic cases. However, circulating AVPc-Abs were also frequently measured in patients with non-idiopathic CDI, demonstrating that AVPc-Abs are not reliable markers of an autoimmune etiology.
Abnormal blood supply: Abnormal blood supply to the posterior pituitary gland was also evidenced in patients with idiopathic CDI, suggesting that selective vascular damage affecting the inferior hypophyseal arteries can be associated with the pathogenesis of CDI.
Vasopressin and its analogue:
Desmopressin (1-deamino-8-D-arginine vasopressin, dDAVP) is the current drug of choice for long-term therapy of CDI. This synthetic analogue has more specific antiduretic action, negligible pressor activity and a longer half life than the native molecule. It can be given parenterally, orally, or intranasally.
The recommended dose of dDAVP is:
100- 1200 μg /day in three divided doses orally;
2-40 μg once or twice a day intranasally; and,
0.1-1 μg parenterally.
There is a large variability in action amongst individuals and hence the duration between doses needs to be determined in each patient.
Dilutional hyponatremia, headache, hypertension and nasal congestion are some of the side effects occasionally seen.
[hr]
Summary:
- Central diabetes insipidus (CDI) is the end result of a number of conditions that affect the hypothalamic neurohypophyseal system.
- The known causes include germinoma/craniopharyngioma, Langerhans cell histiocytosis (LCH), local inflammatory, autoimmune or vascular diseases, trauma resulting from surgery or an accident, sarcoidosis, metastases and midline cerebral and cranial malformations.
- In rare cases, the underlying cause can be genetic defects in vasopressin synthesis that are inherited as autosomal dominant or autosomal recessive.
- The diagnosis of the underlying condition is challenging and raises several concerns for patients and parents as it requires long-term follow-up.
- Proper etiological diagnosis can be achieved via a series of steps that start with clinical observations and then progress to more sophisticated tools.
References:
- Nelson textbook of pediatrics 19th edition
- Horm Res Paediatr 2012;77:69–84 DOI: 10.1159/000336333 Diabetes Insipidus – Diagnosis and Management
- ANURAG BAJPAI, MADHULIKA KABRA AND P S N MENON Central Diabetes Insipidus: Clinical Profile and Factors Indicating Organic Etiology in Children
- Archives of Disease in Childhood, 1986, 61, 247-250, Oral desmopressin in central diabetes insipidus
- Madson Q. Almeida1 Idiopathic central diabetes insipidus: the challenge remains
- Gail E. Richards, MD, Michael J.Natural History of Idiopathic Diabetes Insipidus
[hr]
Submitted by:
Dr. Sudeep Shrestha
Pediatric resident
Institute of Medicine

MD Pediatrics and Fellowship Neonatology, he chooses to stay anonymous. He often writes his views online as well as share few important topics for medical students, doctors and specially parents. He does research in pediatrics.