A) INTRODUCTION
Empyema (aka Empyema Thoracs or Empyema of the chest) is an accumulation of pus in the pleural space that occurs when an infection spreads from the lungs. It comes from the Greek word empyein, which means : pus–producing (suppurates). Empyema itself is not a disease but it is actually a condition complicated by another disease.
B) ETIOLOGY
- Lung diseases:
- Pneumonia (the most common cause)
- Lung abscess
- Tuberculosis
- Rupture of subdiaphragmatic or liver abscess in the pleura
- Post traumatic
- Post-operative
- Blood spread
- Iatrogenic
C) ORGANISMS
The most common:
- Staphylococcus aureus (90% of causes in infants & children)
- Streptococcus pneuomonie
- Haemophilus influenzae
- Group A Streptococcus
- Gram negative organisms
D) EPIDEMIOLOGY
- Most frequently encountered in infants and preschool children.
- Empyema have an incidence of 3.3 per 100,000 children
- Common in winter and spring due to their infective origin.
- Occurs in 5-10% of children with bacterial pneumonia and in upto 86% of children with necrotizing pneumonia
E) AMERICAN THORACIC SOCIETY CLASSIFICATION OF EMPYEMA
STAGE 1 : Exudative with swelling of the pleural membrane
STAGE 2 : Fibrinopurulent with heavy fibrin deposits
STAGE 3 : Organization with ingrowth of fibroblasts and deposition of collagen
F) PATHOLOGY
Empyema has 3 stages –
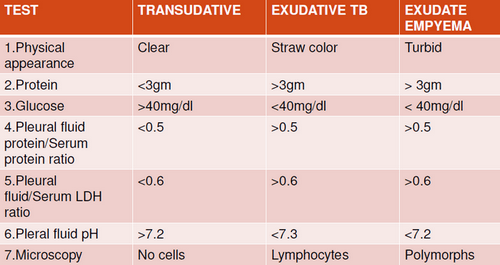
1. Exudative stage: The inflammatory process associated with the underlying pneumonia leads to accumulation of clear fluid with a low white cell count within the pleural cavity (simple parapneumonic effusion)
2. Fibrinopurulent stage: There is deposition of fibrin in pleural space leading to septation and formation of loculation. There is an increase in white cells with the fluid thickening (complicated parapneumonic effusion) and eventually becoming overt pus (empyema)
3. Organizational stage: Fibroblast infiltrate the pleural cavity and the thin intrapleural membrane are re organized to become thick and non elastic (the ‘peel’). These solid fibrous pleural peels may prevent lung re-expansion(‘trapped lung’), impair lung function and create a persistent pleural space with ongoing potential for infection. At this stage spontaneous healing may occur or a chronic empyema may develop.
G) PATHOPHYSIOLOGY
- The pleural space normally contains 0.3ml/kg body weight of pleural fluid
- There is a continuous circulation of this fluid and the lymphatic vessels can cope with several 100 milliliters of extra fluid per 24 hrs
- However, an imbalance between the pleural fluid formation and drainage will result in a pleural effusion
- In health, pleural fluid contains :
- A small amount of cells (mainly mesothelial cells, macrophages, lymphocytes)
- Low protein concentration (0.1 g/l)
- Large molecular weight proteins such as lactate dehydrogenase (LDH)
- Higher levels of bicarbonate ,low levels of sodium and similar level of glucose as compared with the serum.
- These parameters are altered when disease processes such as infections affect the adjacent lung or vascular tissue and activate an immune response and pleural inflammation
- Increased vascular permeability allows migration of inflammatory cells (neutrophil, lymphocytes, eosinophil) into the pleural space
- The process is mediated by number of cytokines such as interleukin(IL) -1,IL-6,IL-8, tumour necrosis factor (TNF)-α and platelet activating factor-released by mesothelial cells lining the pleural space.
- This results the exudative stage of a pleural effusion
- Then progresses to the fibropurulent stage due to increased fluid accumulation and bacterial invasion across the damaged epithelium
- Neutrophil migration occurs as well as activation of the coagulation cascade leading to procoagulant
activity and decreased fibrinolysis - Deposition of fibrin in the pleural space then leads to septation or loculation.
- Therefore, Pleural fluid pH and glucose level falls while LDH levels increase.
H) SYMPTOMS
- Fever
- Fatigue
- Cough
- Breathing difficulty
- Chest pain
- In severe cases, patient may become dehydrated, cough up blood, greenish-brown sputum, run a fever as high as 105 degree F or even fall into a coma
I) SIGNS
Inspection:
- Toxic look
- Signs of respiratory distress- use of accessory muscles,flaring of ala nasi, subcostal indrawing
- Occassionally,empyema may manifest as a pulsatile swelling over chest (Empyema Necessitance)
Palpation:
- Intercostal tenderness
- Tactile vocal fremitus decreased
- Shift of trachea and mediastinum away from affected side
Percussion:
Dullness to flatness may be found on percussion of the chest.
Auscultation:
Breath sound decreased or absent.
J) INVESTIGATIONS
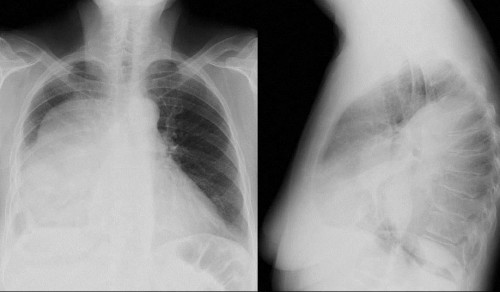
Chest Radiograph:
- Radiographically all pleural effusion appear similar but the absence of shift of the fluid with a change of position indicates a loculated empyema
- Xray flim of chest shows : Shift in mediastinum with obliteration of costophrenic angle and varying degree of opacification
Pleural tap:
May show purulent material full of pus cells
- Blood culture: may show causative agent
- Pneumococcal Empyema culture is positive in 58% cases
- In patient with negative culture results for Pneumococcus, Pneumococcal Polymerase Chain Reaction (PCR) analysis is most helpful for making diagnosis.
- Acute Phase Reactants:
- Leucocytosis is present
- C–Reactive Protein(CRP) increased
- ESR increased
Ultrasound scan of chest:
- Ultrasound must be used to confirm the presence of pleural fluid collection
- It is particularly useful when there is white out lung in chest radiograph.
- Fibrinous septations are visualized
- It is also used to guide thoracocentesis or drain placement.
CT Scan of chest
I) COMPLICATIONS
Complications are common with Staphylococcal infection
1. Local Complications:
- Bronchopleural fistula: If pus is not drained it may dissect through the pleura into lung parenchyma producing bronchopleural fistulas and pyopneumothorax or into abdominal cavity. Rarely the pus dissects through the chest wall i.e. empyema necessitance.
- Purulent percarditis
- Pulmonary abscesses
- Penitonitis from extension through diaphragm
- Osteomyelitis of ribs
2. Septic complications
- Meningitis
- Arthritis
- Osteomyelitis
- Septicemia is often encountered in H.influenzae and Pneumococcal infections
Effusion may organize into thick ‘peel’ which may restrict lung expansion and may be associated with persistent fever and temporary scoliosis.
J) MANAGEMENT
Effective management require:
1) Control infection and sepsis by antibiotics.
2) Evacuation of pus from pleural space.
3) Obliteration of the empyema cavity.
1. Control infection and sepsis by antibiotics:
2. Evacuation of pus from pleural space:
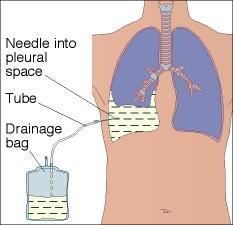
Thoracocentesis: This is a procedure which involves insertion of a needle into the pleural cavity through the back between the ribs on the infected side and a sample of fluid is withdrawn. It is performed under local anesthetics.
Chest tube drainage with or without fibrinolytic agent: Larger surgically placed drains are best inserted in mid axillary line through the ‘safe triangle’. More posterior position may be chosen in presence of loculi. There is no evidence that large bore chest drains confers any advantage small drains should be used whenever possible to minimise patients discomfort. All chest tubes should be connected to a uni-directional flow drainage system (such as underwater seal bottle) which must be kept below the level of patients chest all the time.
Intrapleural Fibrinolytics: Fibrinolytic agent promote drainage, decrease fever, lessen need of surgical intervention, shortens hospitalization. The optimal dose and drug have not been determined. Steptokinase 15000 U/kg in 50ml of 0.9% saline daily for 3-5 days and urokinase 40000 U in 40 ml saline every 2 hrs for 6 doses have been evaluated in randomised trials in children.
3. Surgery:
Indications:
- Failure of chest tube drainage
- Failure of antibiotics
- Failure of fibrinolytics
- Persistent sepsis association with a persistent pleural collection despite of chest tube drainage and antibiotics
Surgical procedures:
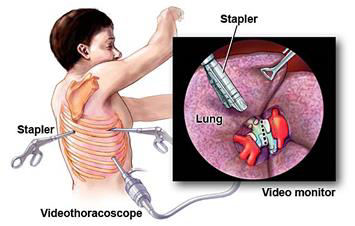
- Video-Assisted Thoracoscopic Surgery (VATS)
- Primary modality for treating complicated empyema after initial therapy
- Higher successful rate(90%)
- Shorter hospital stay, Less cost
- Three port triangle approach
- Achieves debridement of fibrinous pyogenic materials, breakdown of loculations and drainage of pus from the pleural cavity under direct vision.
- Mini Thoracotomy
- Achieves debridement and evacuation similar manner to VATS but it is an open procedure leaving a small linear scar along the rib line
- Decortication
- Involves an open posterolateral thoracotomy and excision of the thick fibrous pleural rind with evacuation of pyogenic material
- It is a longer and more complicated procedure leaving a larger linear scar along the rib line
References:NELSON TEXTBOOK OF PEDIATRICS - 19th Edition GHAI ESSENTIAL PEDIATRICS - 8th Edition MEDSCAPE BTS GUIDELINES FOR THE MANAGEMENT OF PLEURAL INFECTION IN CHILDREN JOURNAL
Submitted By: Kritanjali Ghimire and Monika Shrestha (Intern), Department of Paediatrics, NMCTH