The latest TNM and FIGO staging for Ovarian Cancer has been discussed earlier here. Here, we will discuss pathology of ovarian tumors in short and in a way thats easy to grasp.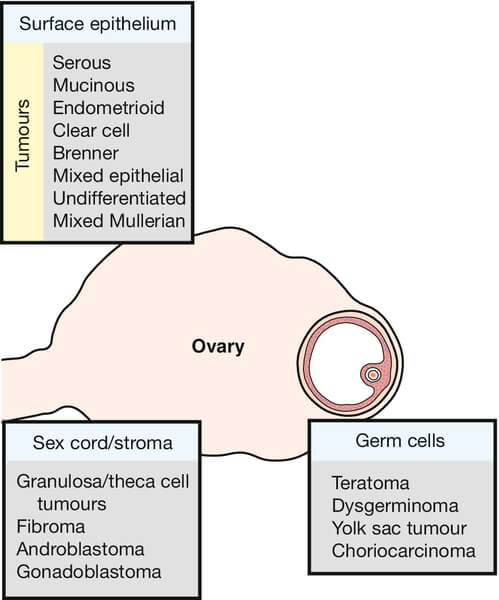
WHO Classification of Ovarian Tumors
| Cells of origin | Surface coelomic epithelium | Germ cells | Sex cord, stromal cells | Metastatic |
| Proportion (%) of ovarian tumors | 65-70 | 15-20 | 5-10 | 5 |
| Proportion (%) of malignant ovarian tumors | 90 | 3-5 | 2-3 | 5 |
| Affected age group | >20 years | 0-25 and >25 years | All ages | Adults |
| Predisposing factors | Hereditary breast ovarian cancer syndrome i.e. BRCA1 (17q) and BRCA2 (13q) mutations
Hereditary nonpolyposis colon cancer (HNPCC) i.e. DNA mismatch repair genes (MLH1, MSH2, MSH6) mutations
Site specific ovarian cancer syndrome
Increased number of ovulation cycles: Nulliparity, Early menarche, Late menopause
Pelvic irradiation
Viral infection (mumps, rubella)
Dietary (high fat, low fiber)
Racial (White, Jewish)
Asbestos and Talc exposure |
Genetic causes
Constitutional chromosomal abnormalities
Dysgenetic gonads |
Peutz-Jegher’s syndrome
Cushing’s syndrome
Meig’s syndrome (Ovarian fibroma + Ascites + Hydrothorax usuallt right sided)
Gorlin syndrome |
|
| Histologic subtypes | Serous
Mucinous Endometroid Clear cell Brenner |
Teratoma
Dysgerminoma Endoderma sinus (yolk sac) tumor Choriocarcinoma Embryonal carcinoma Polyembryoma |
Thecoma
Fibroma Granulosa cell tumor Sertoli-Leydig cell tumor Hilus cell (Pure leydig cell) tumor Small cell carcinoma |
Mullerian primaries: Uterus, fallopian tube, contralateral ovary, pelvic peritoneum
Non-mullerian primaries (Krukenberg tumor): Breast and GIT (colon, stomach, biliary tract, pancreas) |
| Gonadoblastoma (composed of germ cells and sex cord-stromal derivatives) | ||||
| Tumor markers | CA-125: Non-mucinous
CEA, CA19-9: Mucinous
Activin: Undifferentiated |
AFP and alpha-1-antitrypsin: Endoderma sinus (yolk sac) tumor
hCG: Choriocarcinoma
LDH and Neuron-specific enolase: Dysgerminoma
AFP + hCG: Embryonal cell carcinoma, Polyembryoma
None: Immature teratoma |
Androgen: Sertoli-Leydig cell tumor
Inhibin and Estradiol: Granulosa cell tumor
|
|
Pathology of Subtypes of Ovarian Tumors
| Type and Subtype | Proportion (%) | Benign/Malignant | Uni- or Bi-lateral | Characteristic pathologic features |
| Epithelial | ||||
| Serous | 50% | 60% benign
25% malignant
40% of all malignant ovarian tumors and 20% of all benign ovarian tumors |
Bilaterality: 20% in benign, 30% in borderline and 66% in malignant. | Ciliated columnar serous epithelium; +/- papilla; +/- psammoma bodies; tend to be unilocular |
| Mucinous | 25% | 80% benign
5-10% malignant
10% of all malignant tumors. |
10-20% are bilateral | Non-ciliated tall columnar epithelium with apical mucin; tend to be larger and multilocular; pseudomyxoma peritonei |
| Endometrioid | 10% | Majority are malignant
8-15% of all malignant tumors. |
Bilaterality in 25-40% | Non-ciliated columnar epithelium similar to endocervical glands; Synchronous endometrial carcinoma or hyperplasia in 1/3rd cases |
| Clear cell | 5% | Malignant | Bilaterality in 12-40% | Clear cells resembling clear cell renal carcinoma (mesonephros); hobnail cells; Endometriosis in 25%; worse prognosis, poor platinum response |
| Brenner | 2.5% | Mostly benign | Usually unilateral
Can be associated with another epithelial ovarian neoplasm of ipsilateral or contralateral ovary in 30% |
Transitional cells resembling urothelium; Nest of cells; Tend to multilocular |
| Germ cell (GCT) | ||||
| Dysgerminoma | 35-50% | Commonest Malignant GCT | 10-15% bilateral | Ovarian counterpart of seminoma; large cells arranged in alveoli with clear cytoplasm; intense infiltration of lymphocytes and plasma cells in fibrous septa; extremely radiosensitive |
| Endodermal sinus (yolk sac) tumor | 20% | 2nd commonest Malignant GCT | Usually unilateral | Schiller-Duval bodies (glomeruloid bodies) |
| Embryonal carcinoma | Rare | Malignant | Usually unilateral | Primitive embryonal elements |
| Polyembryoma | Rare | Malignant | Embryoid bodies; least radiosensitive GCT | |
| Choriocarcinoma | Pure choriocarcinomas are extremely rare (often found in combination with other GCT) | Malignant | Usually unilateral | Gestational or non-gestational (metastatic from uterine choriocarcinoma); trophoblasts; isosexual precocious puberty is common |
| Teratoma | Immature account for 20% of malignant GCT | Mature/Dermoid cyst and Struma ovarii – Benign
Immature and carcinoid – Malignant |
2-5% immature teratoma are bilateral | Mature/Dermoid cyst (46, XX) – germ cells arrested in 1st meiotic division: Rokitansky protuberance, Unilocular cyst lined by stratified squamous epithelium with underlying sebaceous glands, hair shafts, skin adnexa (ectodermal) +/- cartilage, bone, thyroid, etc.
Monodermal: thyroid tissue (struma ovarii); intestinal epithelium (ovarian carcinoid)
Immature: Fetal tissues derived from 3 germ layers |
| Mixed GCT | 10-15% | Dependent upon cell types present | Dysgerminoma is the commonest tissue element | |
| Sex cord stromal tumor | ||||
| Thecoma | 1% of all ovarian tumors | Benign | Bilaterality is rare | Postmenopausal; Spindle-shaped cells with Oil-red O positive estrogen vacuoles; Endometrial hyperplasia (functional ovarina tumor) |
| Fibroma | 4% of all ovarian tumors | Benign | Bilaterality is rare | Spindle-shaped cells; Oil-red O negative; Non-functional; Meig’s syndrome |
| Sertoli-Leydig cell tumor (Androblastoma); hilus cell tumors are pure leydig cell tumors | 0.5% of all ovarian tumors | Potentially malignant | Usually unilateral (90%); Bilaterality is rare | Testicular cells – sertoli and leydig cells (reinke crystalloids typical of leydig cells); functional (androgens >>estrogen); may stain inhibin positive |
| Granulosa cell tumor | 2% of all ovarian tumors (commonest ovarian stromal tumor) | 5% of all ovarian malignancies | Usually unilateral (bilateral only in 2%, i.e. rare) | Inhibin positive; Call-Exner bodies (like rosette), functional (estrogen >>androgen); tend to rupture (acute abdomen) |
| Metastatic | ||||
| Krukenberg tumor | 5-10% of all ovarian tumors | 30-40% of all ovarian metastases | Commonly bilateral | signet ring appearance; intact capsule; retrograde lymphatic spread (most common gastric cancer followed by colorectal carcinoma) |
Benign Vs Malignant Ovarian Tumors in general
| Features | Benign | Malignant |
| Clinical | ||
| Laterality | Unilateral | Bilateral |
| Mobility | Mobile | Fixed |
| Feel | Cystic | Solid/Variegated |
| Surface | Smooth | Irregular |
| Ascites | Absent | Present |
| Growth | Slow | Rapid |
| Patient’s age | Younger | Older |
| Nodular Pouch od Douglas | Absent | Present |
| Laparatomy | ||
| Ascites | Absent | Present – often hemorrhagic |
| Exophytic growth on surface | Absent | Present |
| Adhesions | Absent | Present |
| Cut-section | Cystic | Solid and hemorrhagic |
| Peritoneal nodules | Absent | Present |
| Ultrasonography | ||
| Solidity and size | Cystic; <8 cm; calcification and teeth | Cystic with solid component >50%; 8 cm; multilocular; bilateral; ascites; peritoneal masses/omental caking; lymph node involvement |
| Color Doppler evaluation | Regular vascular branching and flow | Neovascularization, low resistance flow with pulsatility index <1. |
Patterns of Spread of Ovarian Tumor
1. Surrounding pelvic tissues: Directly
- Fallopian tubes
- Uterus
- Contralateral adnexa
- Rectum, urinary bladder and pelvic sidewall
2. Dissemination beyond pelvis: 3 modes –
a. Intraperitoneal (commonest): Tumors are shed into peritoneal cavity and follow the normal routes of peritoneal circulation.
- Greater omentum
- Right subphrenic region (in supine position)
- Pouch of Douglas (in upright position)
b. Lymphatic:
- Follow ovarian veins (mainly): to the left para-aortic and the right paracaval lymph nodes at the level of the renal hilum and are the most common sites for metastatic adenopathy.
- Lymphatics of the broad ligament: drain into the pelvic lymph nodes, external iliac, hypogastric, and obturator chain.
- Round ligaments: Spread to the superficial and deep inguinal nodes
c. Hematogenous (rare): Choriocarcinoma, embryonal carcinoma
- Commonest: to liver
- 2nd commonest: to lungs
- Others: Brain, bone, kidney, adrenal glands, spleen
Risk of Malignancy Index (RMI)
| Feature | RMI 1 Score | RMI 2 Score (more sensitive) |
Ultrasound features:
|
0= none 1= one abnormality 3= two or more abnormalities
|
0= none 1= one abnormality 4= two or more abnormalities
|
| Premenopausal | 1 | 1 |
| Postmenopausal | 3 | 4 |
| CA125 | U/ml | U/ml |
|
RMI score = ultrasound score x menopausal score x CA125 level in U/ml.
|
||